 Boehringer Ingelheim Pharmaceuticals announced new presentations at the American Thoracic Society’s 2018 annual conference that reinforce the efficacy, safety and tolerability profile of Ofev (nintedanib) in patients with idiopathic pulmonary fibrosis (IPF). “The data presented at the conference support the established efficacy of Ofev, while reaffirming the safety profile observed in the clinical trials and following approval,” said Christopher Corsico, M.D., Chief Medical Officer, Boehringer Ingelheim.
Boehringer Ingelheim Pharmaceuticals announced new presentations at the American Thoracic Society’s 2018 annual conference that reinforce the efficacy, safety and tolerability profile of Ofev (nintedanib) in patients with idiopathic pulmonary fibrosis (IPF). “The data presented at the conference support the established efficacy of Ofev, while reaffirming the safety profile observed in the clinical trials and following approval,” said Christopher Corsico, M.D., Chief Medical Officer, Boehringer Ingelheim.
Ofev, which was first approved in the USA in October 201 and in Europe in January 2015, generated sales of 920 million euros ($1.1 billion) in 2017, a rise of 52.3% on the previous year.
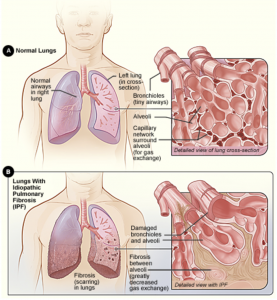
New IPF Mortality Analysis
Pooled data from the two Phase III INPULSIS trials and the Phase II TOMORROW study compared the observed number of deaths in patients treated with Ofev or placebo with the predicted rate of death based on GAP stage over one year.GAP stage is used to predict IPF prognosis and is based on gender, age and lung function (as measured by forced vital capacity [FVC] decline predicted and DLco % predicted). Higher stages of GAP are associated with an increasing risk of death.
Across the population, there were fewer deaths observed in each treatment group than predicted based on GAP stage at baseline. In the Ofev group, the number of observed deaths was 46.7% of the number predicted based on GAP stage, while in the placebo group the number of observed deaths was 63.9% of the number predicted. Based on these differences, the analysis suggests that Ofev may be associated with a 26.8% relative reduction in the risk of death compared with placebo over one year.
“IPF is a progressive and fatal disease, and treatment with nintedanib can slow disease progression by reducing the rate of lung function decline,” said Christopher Ryerson, assistant professor at the University of British Columbia Centre for Heart Lung Innovation, Vancouver, Canada. “Although the individual trials were not powered to measure mortality, our pooled analysis suggests that nintedanib may offer a survival benefit for IPF patients.”
“The symptoms of IPF can have a serious impact on a patient’s quality of life, resulting in a loss of independence and involvement in daily activities,” said Michael Kreuter, Professor at the Center for Interstitial and Rare Lung Diseases, Pneumology and Respiratory Care Medicine, University of Heidelberg, and a Member of the German Center for Lung Research, Germany. “Our analysis observed association between the extent of lung function decline and quality of life. Stabilizing lung function, therefore, may allow patients to retain some of their daily level of functioning, which might improve quality of life,” he added.
For more information, see Boehringer Ingelheim’s press release on these studies.


